We've always been curious: how exactly do neurons in the brain work? Before advanced imaging technology, it was like looking at the world through frosted glass—blurry, superficial, and damaging to specimens. Then a "golden combination" emerged: two-photon microscopy + calcium imaging + head-fixation system, allowing scientists to truly see a living, awake, thinking brain for the first time.
Why Can't Conventional Microscopes See a Living Brain?
Limitations of Traditional Fluorescence Microscopy
🔻 Light illuminates the entire layer → strong background noise
🔻 The thicker the tissue, the less you see → limited imaging depth
🔻 Prolonged illumination damages cells → high phototoxicity
The core requirements of neuroscience are precisely: imaging living tissue, at depth, over extended periods, capturing real activity. Traditional microscopy simply cannot do this—until the advent of two-photon microscopy.
1. Two-Photon Microscopy — The Optical Revolution in Brain Imaging
Core Principle — Two Weak Photons Simultaneously Excite Fluorescence Instead of One Strong Photon
- Conventional fluorescence: 1 high-energy photon → random layer → entire layer fluoresces
- Two-photon: 2 low-energy photons → must strike the same point simultaneously → only then is fluorescence excited
The probability of two photons coinciding in time and space is extremely low (requiring overlap within 10-16–10-18 seconds), occurring only in the minuscule focal region of the laser. This is why two-photon microscopes demand exceptional light sources. The primary laser used today is the titanium-sapphire femtosecond pulsed laser, which delivers ultra-dense pulses at the femtosecond scale, dramatically increasing the probability of simultaneous two-photon excitation at the focal point. The emergence of the femtosecond laser is what made two-photon imaging a practical reality.
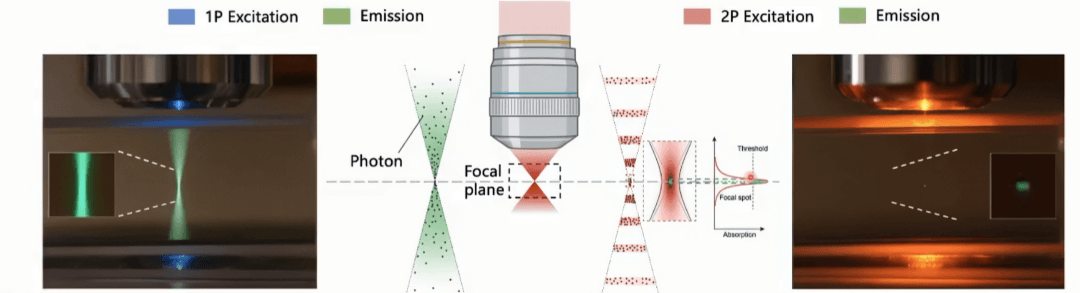
Image source: "1P vs 2P fluorescence imaging" by Steve Ruzin and Holly Aaron
Used for academic educational purposes only. Contact us for removal if needed.
Key Characteristics of Two-Photon Imaging
- Intrinsic optical sectioning: No confocal pinhole needed—built-in layer-by-layer scanning capability.
- Near-zero background: Non-focal regions remain completely dark, producing exceptionally clean images.
- True 3D imaging: By varying focal depth, sequential layers can be scanned and 3D brain structures reconstructed.
Why Does Two-Photon Use Near-Infrared Lasers? (650–1100 nm)
Near-infrared light acts as a gentle deep-tissue probe: less scattering enables deeper brain penetration; lower absorption means minimal thermal damage without harming neurons.
Three Core Advantages of Two-Photon Microscopy
- Deep-tissue imaging champion: Imaging depth reaches hundreds of micrometers, directly observing cortical neurons.
- Ultra-low phototoxicity: Excitation occurs only at the focal point—surrounding cells remain unharmed, enabling continuous imaging for hours.
- Ultra-high signal-to-noise ratio: No background interference; crystal-clear images with maximum contrast.
Significance for Neuroscience
Through "simultaneous two-photon excitation," two-photon microscopy achieves deep, low-damage, high-resolution, live-tissue imaging—the fundamental hardware backbone for observing the brain. It transforms research from static anatomy to dynamic function: watching neurons fire in real time, tracking synaptic connectivity changes, and decoding how neural networks operate.
2. Calcium Imaging — Making Neural Activity Visible as Light
With two-photon microscopy as the hardware foundation, how do we actually see real-time neural activity?
Neurons transmit information via electrical signals (action potentials), but electricity is invisible. Scientists found the perfect proxy: calcium ions (Ca²⁺).
When a neuron fires, calcium ions flood into the cell. By tracking calcium concentration, researchers can indirectly determine: this neuron just fired!
Star Tool: GCaMP Calcium Fluorescent Protein
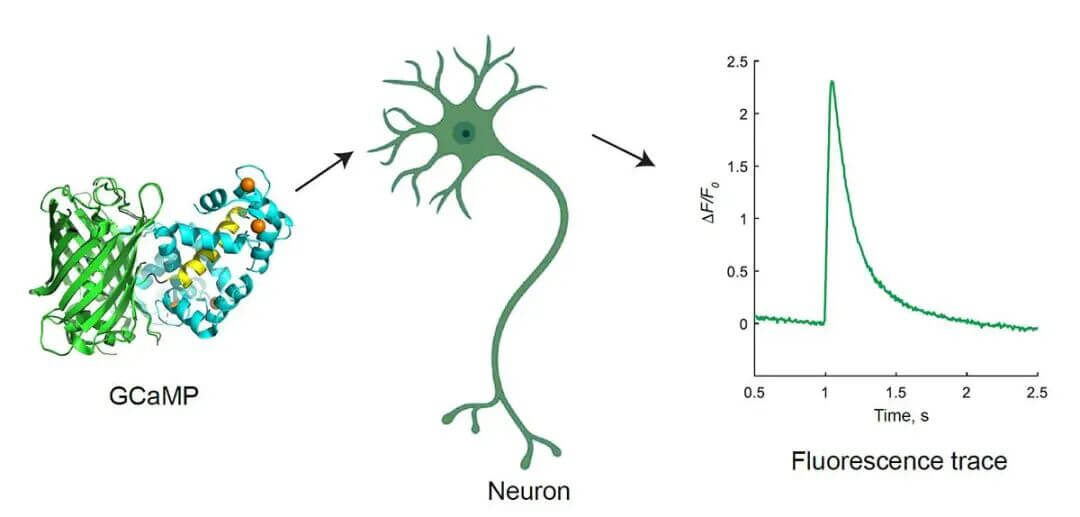
Image source: Yan Zhang, Loren L. Looger, The Journal of Physiology, 22 February 2023
Used for academic educational purposes only. Contact us for removal if needed.
GCaMP is a genetically encoded "calcium sensor light": dark when unbound to calcium, immediately bright when calcium binds. Changes in brightness = neural activity intensity.
An important note: calcium signals ≠ electrical signals
- Electrical signals: ultra-fast, direct, millisecond-scale
- Calcium signals: slightly slower, indirect, reflecting population activity
Although an indirect indicator, calcium imaging is simple, stable, and enables large-scale observation—remaining the mainstream approach today.
Why Must It Be Paired with Two-Photon?
Calcium imaging depends on fluorescence, and two-photon microscopy excels at deep imaging, is gentle on living tissue, and offers ultra-high resolution. Together, they enable simultaneous observation of hundreds of neurons' dynamic activity within a living mouse brain.
Three Classic Experimental Scenarios
- Sensory information processing: Show images or play sounds to mice and observe which neurons are activated.
- Behavioral task encoding: Track how the brain orchestrates actions as mice run or make decisions.
- Learning and memory remodeling: After repeated training, neural activity patterns change—recording the brain "rewiring" itself.
3. Head-Fixation System — Keeping Mice Still for Imaging
The Biggest Challenge: Any Movement Blurs the Image
In vivo imaging is most vulnerable to motion artifacts, but mice simply won't stay still. The solution: design a head-fixation system.
Core Concept: Fix the Head, Preserve Behavioral Capability
Three Core System Components
- Headring: Surgically implanted on the mouse skull, providing a stable "fixation interface."
- Fixation frame: A high-precision mechanical structure that locks the head in place with repeatable positioning.
- Behavioral platform: Treadmills or running disks that allow mice to run and think normally while head-fixed.
Why Is Awake Imaging Essential?
Anesthesia profoundly suppresses neural activity—what you see is not the brain's true state. Modern neuroscience demands data from awake, freely behaving subjects engaged in real activity.
Standard Experimental Workflow
1. Surgery: Implant headring + cranial window
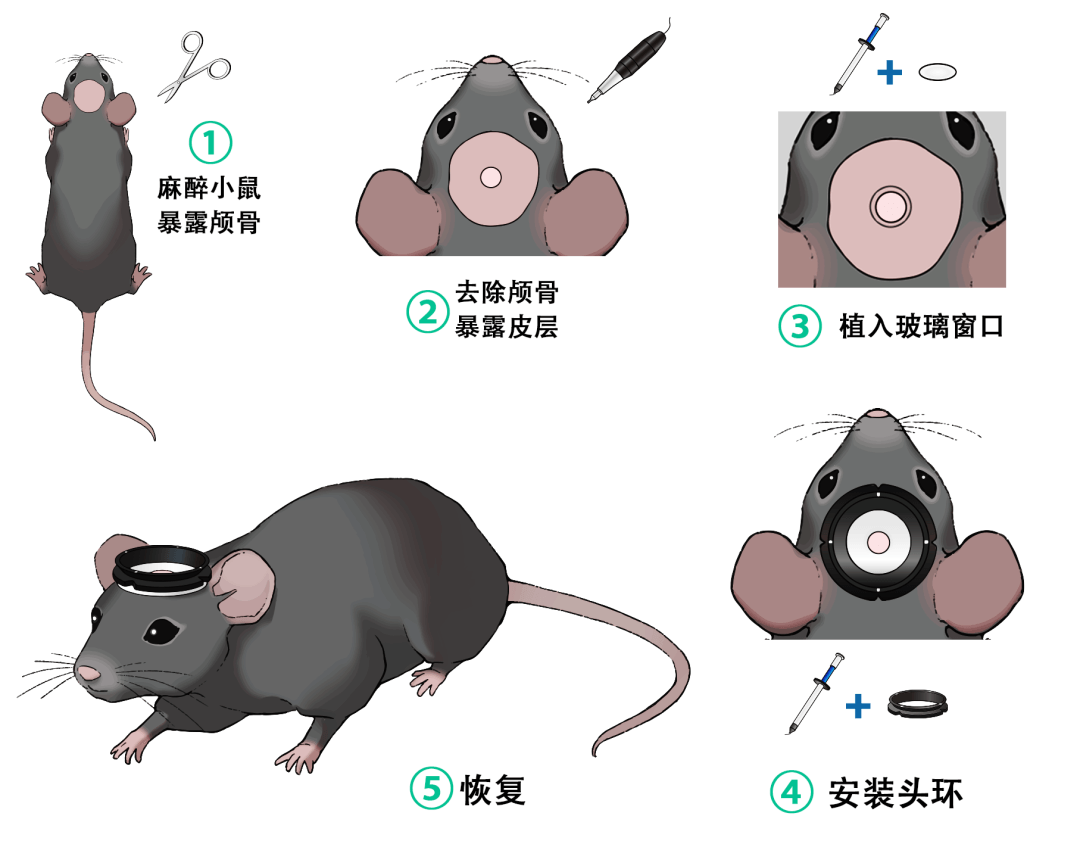
2. Recovery: Wait for wound healing
3. Training: Habituate the mouse to the fixation environment
4. Imaging: Stable recording of neural activity

The head-fixation system completes the final link. Through imaging technology → animal behavior → neural activity, we can study how the brain drives behavior in real time—and through this feedback loop, progressively achieve reverse control.
About Us
SITRANTECH has been deeply engaged in biomedical engineering, collaborating closely with frontline laboratories for over five years. We bridge the gap between "experimental vision" and "engineering reality"—offering both micron-precision standardized in vivo behavioral equipment (such as awake mouse head-fixation systems) and agile custom engineering services to transform your unique research ideas into reliable experimental data. Our mission is simple: empowering scientific discovery through precision engineering, bringing innovation from the lab to real-world applications.
Originally published on SITRANTECH WeChat Official Account









